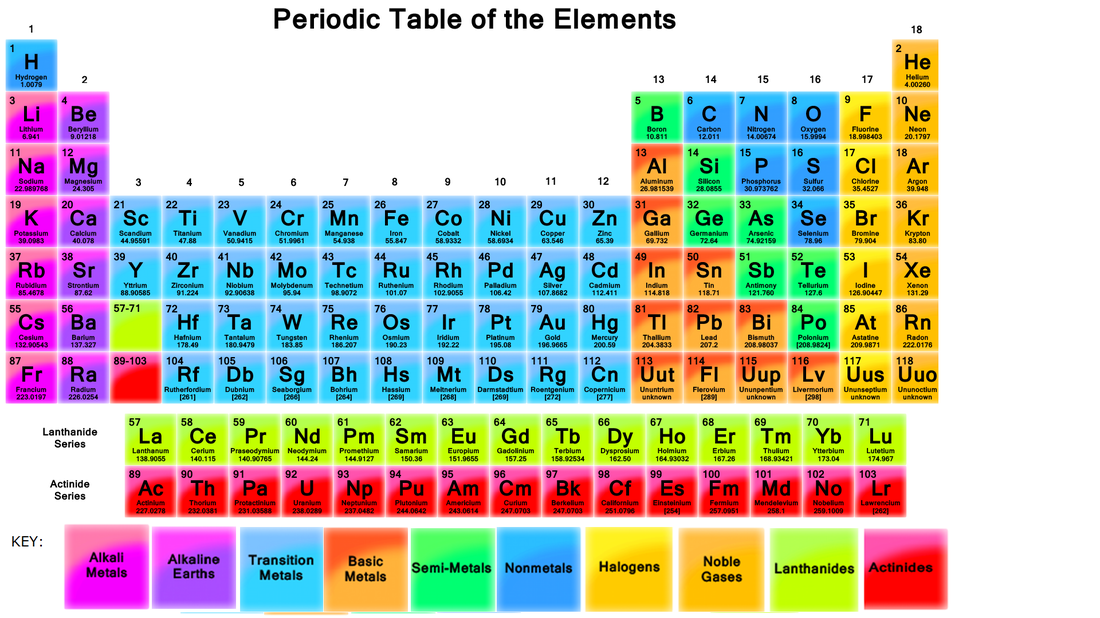
The Periodic Table
The periodic table arranges elements by their atomic number and groups them by their chemical properties.
The periodic table can vary depending on its use, and can include a wide range of information, from its atomic number to its radioactivity. Below is a table containing basic information.
The periodic table can vary depending on its use, and can include a wide range of information, from its atomic number to its radioactivity. Below is a table containing basic information.
http://chemistry.about.com/od/periodictables/ig/Printable-Periodic-Tables/Periodic-Table-Wallpaper.-kIG.htm

The element hydrogen (H) will be constructed from hydrogen atoms.
The element helium (He) will be constructed from helium atoms.
The number in the top left corner of an element, is its atomic number. We can recall that this is the number of protons an atom has.
The Elements name is written below the elements symbol. This is used to identify atoms.
The number below the atoms name is its atomic mass.
The periodic table is one of the most basic tools in the field of chemistry, and will be refereed to regularly. As such it is important to have a thorough understanding of how to use the periodic table.
As the number of neutrons and protons in each atom increases, so does its atomic mass.
The periodic table is broken into a number of categories, as shown by colour, each of which have their own unique chemical properties. The individual properties of each group will be analysed later.